
vv3rtigo
-
Joined
-
Last visited
Posts posted by vv3rtigo
-
-
-
-
9 minutes ago, chenbeier said:
Your photo, I cannot open. 404 file not found on unbuntuu
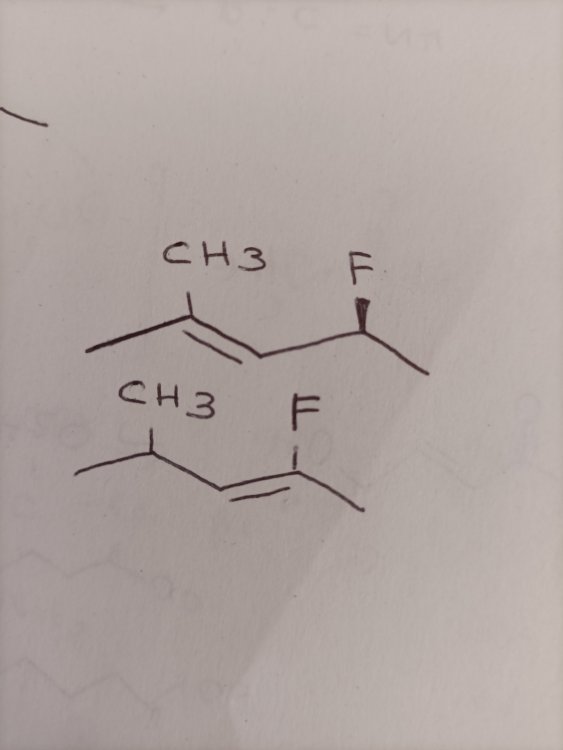
it's an elimination reaction (E2) with NH3 as a solvent. The substrate is: 2-methyl-3-Chloro-4-Fluoropentane.
The products are two alkenes (i put them in the photo but I don't why you can't see it). One of the alkenes has the the double bond on the C2-C3. The other one on the C3-C4.
My professor wanted to know which product is more stable.
-
1 hour ago, exchemist said:
What does "do an E2 with NH3 on the 2-metyl-3R-Cl-4S-F pentane"mean? E2 I assume means an elimination reaction, somehow involving ammonia, but what are the substituents on pentane and what were the 2 products? There seems to be a .jpg file attached which we can't see.
If you use a few more words, it may help to clarify what this is about.
Yes it's an elimination reaction (E2) with NH3 as a solvent. The substrate is: 2-methyl-3-Chloro-4-Fluoropentane.
The products are two alkenes (i put them in the photo but I don't why you can't see it). One of the alkenes has the the double bond on the C2-C3. The other one on the C3-C4.
My professor wanted to know which product is more stable.
-
-

Which alkene is more stable?
in Organic Chemistry
So the one with the Fluoro isn't stable?