-
Posts
3887 -
Joined
-
Last visited
-
Days Won
1
Content Type
Profiles
Forums
Events
Everything posted by Enthalpy
-

Neanderthals Built a Water Reservoir
Enthalpy replied to Enthalpy's topic in Evolution, Morphology and Exobiology
Other speculative uses for the construction. Access may have been easier then, before sediments accumulated. ========== A warm bath or a sauna, using hot stones maybe. In a cave, less cold than outside during that ice age, and far from wild animals when the users took their shoes off. Deep in the cave because water dropped there. The constructors could also wash their clothes and shoes there. If having only one set, they would cherish the safety and relative warmth. ========== A pot to cook vegetables. Meat can be grilled, some nourishing vegetables can't. Manioc, American pea grow naturally in South America (I didn't search for European examples) so cooking can predate agriculture. Why so deep in the cave? Water available there, like one litre per person and per meal, saves only a feasible transport effort. The location protects also the meal from competing humans and animals. ========== A pot to tan animal skins with bark to make rot-proof leather? Yes, this is technology. But they attached sharp stones to a shaft or handle, which needs more invention and effort. Again, running water is a comfort, protection by the cave is useful. But imagine the constructors hunted sheep-sized game. Two animal skins dress one adult Homo. The 50kg meat feed that human for 100 days, a bit more with vegetable side dishes. So people could replace the animal skin as these rotted, with some sewing effort. ========== A grill? If wood was scarce then, the constructors would have burned grease, better in a pot. A cave protects against wind, cold, animals - but why so deep in the cave and where water drips? ========== A site to cut up the hunted animals? Separate the meat, skin, bones, tendons, guts takes times, cleaning takes more. You don't do that in the living room, outside there were animals and it was often cold. Running water seems necessary, better deep in the cave than at the river. Drawbacks: such sites may already be known, not in caves, and for big game, much is done before the transport. ========== I still prefer the reservoir of reliable drinking water, as it answers a vital need. Marc Schaefer, aka Enthalpy -
Broken after 51 days, that is 76h practice. Redone in 5mn, but durable would be better.
-
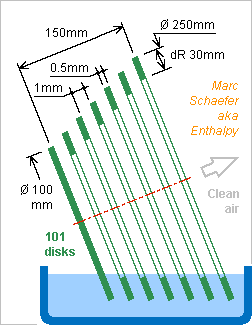
Hello everyone and everybody! Sars-Cov2 allegedly propagates through air as an aerosol. I propose to flow air between many sheets that catch the droplets falling on them. A liquid wetting the sheets shall destroy the viruses and evacuate the residues as the slowly rotating sheets dive in a tank. The liquid's flamability, toxicity, odour, vapour pressure, foam... matter. Maybe some soap or nonvolatile alcohol fit. The sketched example lets air flow radially to the hollow centre where a non-displayed fan extracts it. The sheets should remove most dust before it reaches the fan, and the liquid wash the dust away, so maintenance would mainly replace the liquid. The sheets are tilted so they can pass through water and dust falls on them. This works also at the sides and lower parts of the sheets (because the air flow is parallel to the sheets), advantage over a cylindrical stack of frustums. A few (not displayed) combs can hold the sheets. The example dimensions are not optimized and not displayed on scale. D=10µm droplets fall through air with 2.9mm/s, so 22.5° tilt lets cross 1mm sheet separation in 0.9s, while Brownian motion wouldn't suffice wikipedia - wikipedia dR=30mm allow 33mm/s between the sheets. 100mm cumulated separation, D=100mm to 250mm, and estimated 3/4 turn exposed to air let 1.4L/s flow through. This exceeds a person's peak inspiration during moderate activity (cashier, computer work...), so the cleaned air makes a bubble around the person's head. A single small device processes 80m3 in a 16 hour night, that's an individual office. Operation is very gentle and silent. Air is extracted with only 50mm/s on D=190mm. Laminar flow drops 0.2Pa at the sheets, so the fan needs >0.6m/s near its hub, for instance 150rpm. Slow sheets are quieter, but if they give the rotation speed, then a static flow straightener might replace the fan, possibly with a centrifugal flow, hi Tesla. Operation needs very little power: 0.3mW air flow between the sheets, 2µW air acceleration, maybe 0.2mW liquid movement. Losses and decorative Leds consume more. A Usb port, a battery, an AC adapter suffice. Marc Schaefer, aka Enthalpy
-

Neanderthals Built a Water Reservoir
Enthalpy replied to Enthalpy's topic in Evolution, Morphology and Exobiology
Fun: bone powder is sometimes included in clay to make ceramic. fr.wikipedia (in French, sorry) More generally, organic materials strengthen ceramics fired at too low temperature. Please double-check, I know zilch about pottery. ========== Hi Mark Gregson, thanks for your interest! -

Neanderthals Built a Water Reservoir
Enthalpy replied to Enthalpy's topic in Evolution, Morphology and Exobiology
The researchers saw fire traces on the construction in the Bruniquel cave. As cave explorers, they naturally suggest "light" as the reason to make fire, just as, as a backpacker, I naturally suggest "drinking water" as a reason for the construction. The location is near a rain zone, not where I'd put lighting fires. The fires are also about knee-high, which is a drawback as a light source. This leaves many dark zones in the cave, and it dazzles the users. Light sources sit better above our heads, as in our houses, to illuminate the ground where we walk. The planners and builder of the construction could have attached lighting fires high at stalagmites. Was the construction made to keep fire? Bushmen keep fire using pieces of wood like cigars, and the antique Rome had temples to keep and distribute fire, easier than igniting it. The construction was far from wind, animals and bad weather, it had walls to keep oil or fat, and wicks at the sides, where we see char. But I'm not convinced. Why so deep in the cave? Why so huge? Leave the fire in spring, find it in autumn? Why a location where water drops? I prefer my water reservoir theory. Deep in the cave for year-round supply, under water drops, proper size. Clay or similar made a water-tight wall, fire hardened clay, we see burnt stalagmites where clay was thin or absent. But how to test this? Maybe the calcite layer deposited years or decades after the construction contains evidence. It would be very thin: 175mm in 175 000 years make 1µm/year. The analysis methods may detect atoms, possibly isotopes, rather than compounds. Stronger clay concentration in the calcite layer would suggest the constructors brought it on the stalagmites. A composition not found nearby would suggest the constructors brought clay from an other location. A comparison with stalagmites elsewhere in the cave is less sensitive to more recent floods. Would isotopes tell if clay was brought in the cave? 14C fails at 175 000 years. I hope someone finds better tests. Marc Schaefer, aka Enthalpy -

Neanderthals Built a Water Reservoir
Enthalpy replied to Enthalpy's topic in Evolution, Morphology and Exobiology
Ramblings by a non-expert here. I just wonder how many Sapiens sapiens groups lived in Europe unnoticed from present science. Remains from about 300 Neanderthalensis have been found in Europe. If 1 Homo in 300 was a Sapiens sapiens, chances are fair that none has been discovered up to now. And if one is discovered some day, maybe archaeologists will re-attribute some remains that comprise a single phalanx. How many Neanderthalensis populated Europe is unknown. Experts cite figures like 70 000. If the subspecies inhabited Europe for 200ky and individuals lived for 20y as a mean, then 700 000 000 individuals lived on the continent over the period. The Sapiens sapiens could amount to 2 000 000 over the period, without any remains discovered. I just find it hard to imagine Sapiens sapiens sitting on the now-Marocco coast for 250 000 years, seeing Europe, and not crossing the straits. But maybe their mentality, culture and technology differed so much from now. -
Floats down to 2000-3000m sometimes use a "syntactic foam" made of microballoons in a polymer matrix. Microballoons are small hollow glass spheres, the polymer is often epoxy. The density varies between 400 and 600kg/m3 depending on the maximum depth. I propose to mix microballoons of very different sizes to reduce the density. Usual slurries must contain 40-50%vol liquid to flow and prevent voids, but concrete mixes particles of very different sizes to use less water+cement. Similarly here, smaller microballoons would fill much of the volume between the bigger ones so the polymer matrix fills less volume and weighs less. Microballoons for model hobbyists exist in varied sizes. A ratio of 10 is good, so the usual grain sizes 100-300µm and 2-4mm could serve. If available, a third size would save more mass. And as is known, a runny resin simplifies the use of microballoons. The few oil industry papers I read don't mention this combination, but model hobbyists are inventive. ========== Whether the syntactic foam operates deeper? If the spheres have much thicker walls, the composite doesn't float any more. Alternately, the epoxy can be removed, and the bigger spheres be of alumina, then you have Nereus' design, but it was lost at depth. I feel my proposal with lithium intrinsically safer against pressure. Mixing different sizes of microballoons can serve beyond floats. Marc Schaefer, aka Enthalpy
-
A liner of corrosion-resisting metal, for instance nickel, tin... would protect lithium against water. Maybe a simple displacement reaction deposits the noble metal on lithium. The process would drop and turn the lithium part in a solution of the noble metal salt. Water won't fit, but polar solvents without O-H bonds serve in lithium batteries. "Lithium metal" batteries (search words) are a fashionable research topic. Carbonates seem less favoured, but dimethylsulfoxide (DMSO) and dimethylacetamide (DMA) are considered among many more. Both nickel chloride and lithium chloride are well soluble in DMSO, as mere examples. The noble metal layer deposited by the displacement reaction is thin, but once the lithium part is easier to handle, other processes can deposit a thicker liner, of metal, alloy, organic materials like polymers... Marc Schaefer, aka Enthalpy
-
Ahum. (1) Write (2) Read (3) Update. Ta and Nb are not electrodeposited from aqueous electrolytes. Alas, aqueous is the usual choice for Ni, Co, Mo, etc. W is not deposited alone, but its alloys are. Re is deposited. With close metallic atomic radius, Re could be an alternative. Mn is commonly electrodeposited.
-
Many more elements and alloys can be deposited than I had guessed. Here are composition examples from one paper: Fe base material in that paper Ni 80% Co 6% and 95% CoNi 65%+12% V 1% from ammonium metavanadate Cr 10% from banal salts B 8% from KBH4 basic solution If I extrapolate boldly from Fe-based to Ni+Co and Ni-based alloys: Cr may be possible and it lets Ni superalloys resist corrosion better. 20% Fe is feasible and serves in some Ni superalloys. 1% V is a useful hardener in Ni too. But the hardener B uses here a basic solution uncommon with the other elements. ========== Fascinating too: electroform maraging steel Fe-NiCoMo. It would include the precipitation-hardener Ni+Mo but lack the important Ni+Ti. Whether Ta can replace Ti? And could Co-20Cr-16Ni-7Mo-etc be electroformed? It combines fantastic corrosion resistance with good room-temperature strength. The standard cites also 2% Mn and <1.2% Si. ========== Maybe gas turbine blades or complete blisks can be electroformed. Machining hollow blades takes time, spark-machining blisks too, electrodeposition can be cheaper. Heat exchangers need intricate shapes with weld seams, solder seams or difficult machining. Superalloys would resist heat, corrosion and pressure. Electroforming would avoid assembly seams and difficult or impossible machining. Maybe a complete exchanger can be electroformed at once, or the tubes, plates... be made in a separate process or step and the manifolds deposited on them, possibly bored thereafter. Adolphe Sax patented welding by electrodeposition. Marc Schaefer, aka Enthalpy
-
Some prospective thoughts about electroformed hard nickel alloys and conductive hard copper alloys (for combustion chambers) there: scienceforums
-
Some prospective thoughts about electroformed hard nickel alloys (for keys, body parts) there: scienceforums
-
Bassoon players, and oboe players supposedly too, use a reed reamer to fit the bore on the end of the conical bocal. My bocal's end diverges by 1:50 or 1.15°. Reamers sold for that purpose (with a handle on the photo) diverge much more strongly, to fit varied bocal diameters I guess. Consequently, the reed fits only the bocal's tip and is wobbly. I bought for 22€ a used conical reamer for mechanical workshops. 1:50 is a standard, but 1.5° did the job: the difference is 50µm over 8mm fitting length, which the cane absorbs. The reed doesn't wobble any more. A handle isn't mandatory, but mind your fingers with a new reamer. My bocals are D=5.20, 5.30 and 5.50mm wide at the tip. The reamer starts with D=5.50mm, its nominal diameter, not 0.2 or 0.3mm narrower as sources claim. The narrower bocals perceptibly hit the bore's end and roll more easily in the reed, so I may try a D=5.00mm reamer. At 1:50 diameter slope, a 0.3mm narrower reamer plunges 15mm deeper in the reed's throat, which must change the sound a lot. One could also have one reamer per bocal, possibly grinded to the desired diameter, but one reed wouldn't fit all bocals. Or better, all bocals could bear extra thickness at the end, machined to a cone with bigger standard slope and diameter. I have one such bocal. Marc Schaefer, aka Enthalpy
-
I understand photovoltaics as the conversion to electricity first which is a waster of money and efficiency if heating is the goal. In that case, battery. Tesla claim >94% on a cycle and guarantee (by replacing for free) 10 years operation. But if sunlight shall heat a storage medium, I prefer this over a battery, sure. The only decision is whether the heat storage medium shall be hot, then smaller but with an oriented sunlight concentrator, or warm, then bulky but with a static collector, say on the roof's slope.
-
Everything degrades in a laser diode, including the metal conductors and their contacts with the semiconductor, the drift of varied species (including the metal) into the semiconductor, and so on. Expect the laser diode to fail completely soon after it begins to degrade. Not every laser diode has a factor-of-four between threshold and normal operating point, but even then, if you half the current, you half the efficiency too, already through the non-lasing recombinations. Plus some other effects, like the natural emission not matching well the resonator.
-
If the figure-of-merit really is the energy stored at identical volume, and electricity must be involved, a battery is better than heat storage. But I'd no go through electricity, as the conversion from sunlight is expensive and inefficient.
-
I was the project leader for Sara, at the club Esieespace space.skyrocket - digitalcommons.usu.edu Microsats are defined by their mass, but this relates loosely with the capabilities of a satellite, as electronics gets smaller. You need area, which converts indirectly to mass, for instance to broadcast TV from geosynchronous orbit. Big antenna, big solar arrays for power. Unless many satellites can do the job from a low orbit, with little power and less directional antennas, this needs a big heavy satellite. Or for radar imagery. Where you want only sensors, signal conditioning and processing, small satellites are as good as big ones. Just like a mobile phone fit in a luggage piece in 1980 but in a pocket now. I see a different criterion: complexity. Difficulty of design is the first cost factor at one or few satellites (maybe not at 1000), bad company organisation is the second one (or in the reverse order). Size has little to do here. Build the satellite too big, and its launch will cost more. Miniaturise the same complexity, and design cost skyrocket, provided the project finishes. So you must distinguish between a small company, a university team... that defines a simple project and packs the (real, not academic) competence at one place, and a pool of multinational companies where incompetent managers define a standard satellite because they can only copy what they read elsewhere, and order to make it micro because it's fashionable - their conclusion will inevitably be "we tried, and experience tells it's a bad option". A few options that define the complexity and the probability of failure: Has the team already made projects together and with the own hands? Space isn't that difficult. Technology is, organisation too. Has the team already thrown quality standards and methods over board? Do the people experiment and prototype? Can the designers build the parts and equipment themselves? Could you suppress the international or multi-university collaboration? Do you need orbit control? Do you need attitude control? Do you need several ground stations?
-
Passing over electricity is the wrong start to heat from sunlight. Melting paraffins are the best heat stores at identical mass and near-best at identical volume, and they do it at a useful temperature. But is the volume the true goal? Running multiple long air pipes in the ground stores heat in soil, soil costs nothing, and this provides cool air in summer too.
-
With properly built components, my educated guess (my first profession) is 10 years for a LED at normal operating point, more if you derate. At I/5 you still get light, possibly you get 10* more life. And yes, temperature matters a lot, more than the current. For a laser diode it's much less: 1 years continuous operation is already difficult. Alas, you can't derate a laser diode, because it won't lase at all if you reduce the current, even /3. Try the current form factor if possible. Cooling better would bring a lot but it's already optimized.
-
I suggested repeatedly to electroform Ni and Ni-Co, possibly allied with Mo, for music instruments and more scienceforums and later and elsewhere Strong electroformed alloys would serve many more uses. For instance rocket chambers are of nickel electrodeposited on an inner copper jacket where cooling channels have been milled. But what alloys might be possible? Please remember I'm not reliable on electrochemistry. Strong nickel alloys are known, mainly for superalloys used in gas turbines. They consist of Ni, 20% Cr, 0-20% Fe, <20% Co, <10% Mo, <5% Ti, ~1% Al, Nb, and some more. Nice source: Nickel and Its Alloys, monograph 106 by the National Bureau of Standards Based on redox potentials, which are not the whole story for electrodeposition, Co (-0.26V) is fully compatible with Ni (-0.28V), and both are indeed codeposited routinely. Mo (-0.20V) looks easy, and Ni-Mo compound precipitation knowingly hardens Ni alloys without embrittlement. Mo is a very strong candidate. If Cr (-0.91V, forms an oxide layer) can be co-deposited, fine! Zn (-0.76V) is commonly deposited together with Cu (+0.52V) to make brass. My understanding is that Cr protects Ni alloys against corrosion as it does for steel, but isn't a vital hardening element. Since Ni and Co alloys resist corrosion enough at room temperature, Cr can be dropped. Maybe Fe (-0.45V) can be co-deposited. It's present in some Ni superalloys, not in others. It must reduce the density and the cost, little more. Easily dropped. Can Ti, Si, Zr, C, B, Al (-1.67V) be electrodeposited? I suppose not, based on their solubility, potential and oxide layer. Precipitates of Ni-Ti and Ni-Al compounds are very important hardeners of Ni superalloys, but ciao. C is undesired, Si doesn't seem useful. B is sometimes used in tiny amounts in superalloys, adiós. Nb (-1.10V), or rather unseparated Nb+Ta, precipitates as Ni-Nb to harden superalloys. I suggest to try Ta (-0.60V) instead: same atom size, same effects in alloys. If deposition works despite the oxide (it does for Zn), electrodeposited Ni and Ni-Co have a second precipitation hardener. W is sometimes added in tiny amounts. Re (+0.30V) could be a candidate equivalent. ========== Cu-Zn brass is routinely deposited. Cu-Ni-Zn "nickel silver" must be about as difficult and is the common alloy for keyworks of woodwind instruments. Though, I want to make keys hollow and thin to be lighter and stiffer. Bigger diameters help, stronger alloys too, as inspired by nickel superalloys. Lighter keys would be very appreciated at the German bassoon, at other instruments too, lighter and stiffer keys at the baritone saxophone. ========== Since I'm here: the cooling jackets of rocket engines mention exotic Cu alloys meant to conduct well and be harder than pure Cu. The widely available Cu-Cr1Zr was developed for that purpose, along with few more. For electrical conductivity, but it's the same. Marc Schaefer, aka Enthalpy
-
I eventually checked what encompass the LCP polymers I suggested for instrument parts. LCP (liquid crystal polymer) is a very broad class of polymers that includes varied chemical families and properties, some adequate here, others not. Instrument parts need thick raw material, not just fibres, and they need stiffness, possibly after stretching. Some of the thermoplastic LCP can be injected, say for brass and single-reed mouthpieces, but luthiers use to shape parts by turning and milling rods up to now. Vectra, from Ticona-Hoechst-Celanese-Kururay-andsoforth, is just one seducing LCP. Vectran is the name as a fibre. A950 is one unloaded Vectra. tools.celanese filter LCP and Vectra There are dozens LCP more, some fit the present use, others like aramides exist only as fibre. One list of LCP there plastics.ulprospector Many exist only with fibre load, which uses to blunt the chipping tools, so I guess stretching the pure polymer for stiffness is better. Stretching in one direction, say L, expectedly loses stiffness along R and T. So what anisotropy is acceptable? Take a body joint with ID=14mm OD=24mm L=300mm and arbitrary E=10GPa rho=1300kg/m3. The elliptic deformation resonates at 28kHz and bending at 0.76kHz, so EL*37 and (does it?) ET/37 would bring both to pleasant 4.6kHz. Though, A950 fibers near "only" 100GPa, so the cold or lukewarm stretch can be strong if not embrittling along R and T. Work-hardening must also ease the subsequent chip machining, up to some hardness limit. I got vague hints to the price of Vectra A950 pellets 10-25usd/kg Alibaba 55usd/kg Alibaba while a fabric is logically more expensive 100€/kg fibermaxcomposites Hoechst-andsoforth can extrude pellets to bars. But how expensive is grenadilla Dalbergia melanoxylon? Openly available data is even less accurate: supposedly for lesser quality but for tiny customers 60usd/kg rarewoodsusa So cornettos, tárogatók and all woodwinds except the lower saxophones can afford equally Dalbergia or Vectra. But there are more elements of comparison Dalbergias cross borders uneasily and get rare, big parts more so. Most LCP are insensitive to moisture, wow. Temperature does little to LCP, not even expand it. Dalbergia needs decade(s) drying in near-net shape at the luthier. Polymers let try new instrument shapes immediately. Some polymers are stiffer than Dalbergia. Marc Schaefer, aka Enthalpy
-
To scrape a bassoon reed, a plaque inserted in the thin cane supports it and makes it convex. My plaque is made of bone. The material offers the proper hardness to be filed or sanded at decent pace and resist accidents when scraping the reed. This seems better than the usual plastic or metal. Sand paper doesn't let bone dust stink like machine tools do. My raw material was already a commercial part, so I can't tell what bone of what animal it was. Ask the butcher. Marc Schaefer, aka Enthalpy
-
Cornetto by Lene Langballe, with music and explanations: UC3F8lha4UTF-2U2GHdceMCg
-
Science for music instruments is a debated issue, mainly because acousticians make consistently bad instruments. They don't find explanations for what musicians perceive, neither, and some even claim the perception is wrong (wall material at wind instruments). Still now, the best way to design an instrument is empirical, with or without theoretical explanation. You can view this optimistically: the acoustics of music instruments is one domain where science can and must progress. With ideas, experiments, but needing little maths and money. A bizarre aspect is that many luthiers are indeed excellent scientists, with theoretical knowledge, willing to experiment and improve the instruments. From the patents, some of them have more knowledge on theoretical acoustics than academic research has, but they don't share this knowledge - they write "we found by chance that...". Music instruments evolve slowly because they're poorly understood (reading research papers gives me the impression of being the frontrunner), and because finding anything better is difficult (I started describing some here in 2017 and had begun thinking decades before), and because musicians do have legitimate reasons for conservatism, after 20 years learning an instrument. And nevertheless, the piston trumpet, the Boehm flute, the German bassoon... which needed to learn a new instrument, were all adopted. Not immediately by everyone, but soon and fast enough that one or more luthier could live from that. So I have good hope that my proposals get adopted, when the present instrument isn't satisfactory. That's the case for the bassoon and the cornetto at least, maybe the oboe. The flute is less clear.
-
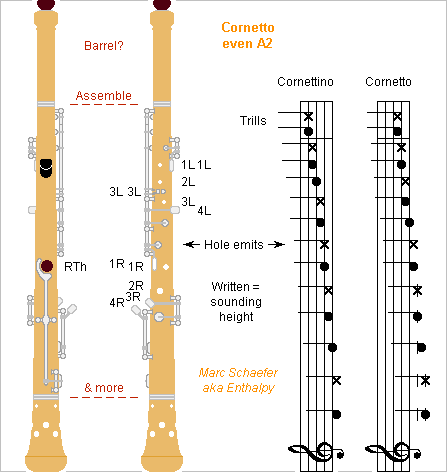
From rare first-hand information, the cornetto plays lower and is longer than I thought, so here's my cornetto with even fingerings, version A2, and its possible aspect: The beautiful and loud cornetto has no modern equivalent. I hope a version easier to play will help revive it. This system A2 shifts to correct height the system A already described here: all holes are independent and any note opens all holes below the main transition, so regular hole diameters and distances achieve even intonation, timbre and emission. The few keys allow many designs. I eased the orientation of the very low right hand. As previously, one individually controlled tone hole per semitone makes a convenient chromatic instrument and enables optimum fingerings for easier high registers. Arrows indicate the lone open holes. Real life and narrow holes may let the fingerings differ a bit from the ideal case depicted here. Both index could operate only one hole, if the thumbs operate three, helped by hand rests. Better hand positions, less simple keyworks. If the usual high notes get easier with my system, slightly broader bore would ease the low notes and stabilize their height. As the cornetto's sound is mellow, reasonably broader tone holes would stabilize the intonation, with more covers if needed. Inspired by a clarinet, not a flute nor saxophone. High tone holes narrower than the low ones let losses match at register jumps and make lone open holes more efficient. The bore profile, which may need a barrel, must adjust the intervals between the registers. Rounding the hole edges at the finger and column ends should improve much. If any necessary, chambers at the tone holes can mellow the sound. Multiple holes increase the losses at identical inductance, oblique they mellow the sound. Small holes added at the longer bell even out the lowest notes. scienceforums Marc Schaefer, aka Enthalpy